科研焦點
(只有英文版本)
Title: |
Professor Baoting ZHANG from School of Chinese Medicine, CUHK develops a new strategy for virtual screening of natural products targeting microRNA-mRNA interaction |
A research team led by Professor Baoting ZHANG from School of Chinese Medicine of the Faculty of Medicine at The Chinese University of Hong Kong has developed a novel drug discovery strategy for the virtual screening of natural products for microRNAs (miRNAs). The findings of the research were recently published in the world-renowned journal Advanced Science (Impact Factor: 15.840) (https://onlinelibrary.wiley.com/doi/10.1002/advs.201903451).
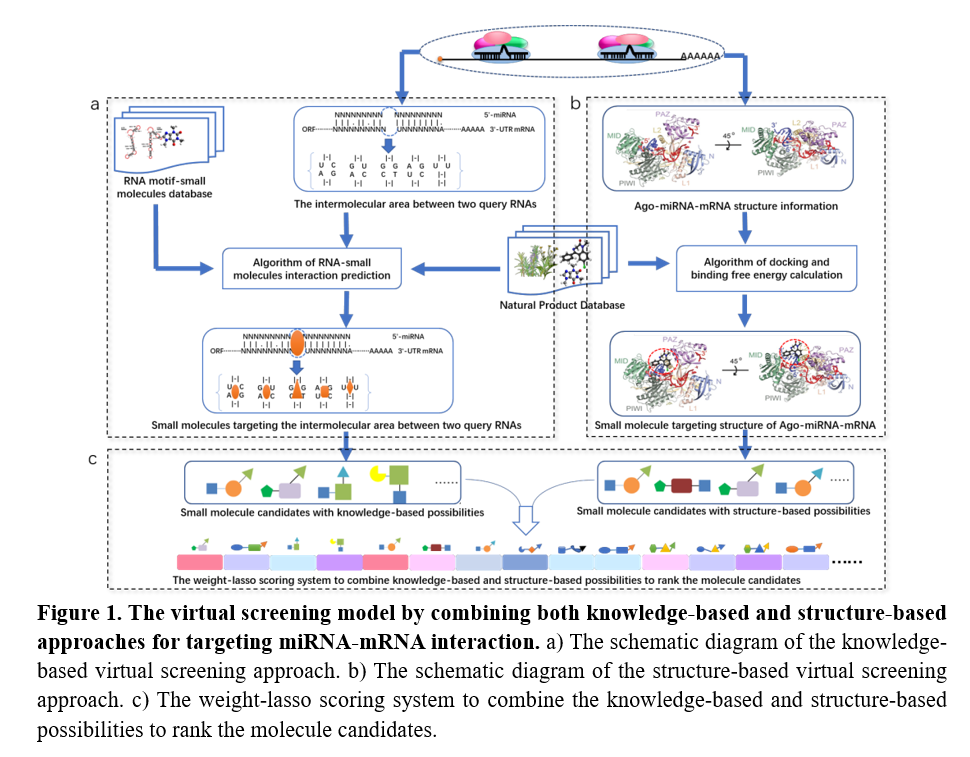
The miRNAs are small non-coding RNA molecules with a length of 21 to 23 nucleotides that are widely present in eukaryotes and can regulate gene expression. Many studies have confirmed that miRNAs play important roles in various pathophysiological processes. Professor Baoting ZHANG said: “Through the analysis of miRNA databases of different species, our research team found that miRNA and mRNA can form a unique loop structure during the interaction process. This loop structure is very close to the spatial position of the Argonuate (AGO) protein it guides. The complex composed of miRNA, mRNA and AGO protein is a promising drug target. The research team proposed to develop a virtual drug screening database for miRNA targets. Traditional Chinese medicine and natural products is selected as the source of small molecule compounds due to their advantage of structural diversity and biological activity. Then we applied the above virtual screening strategy to carry out high-throughput virtual screening and experimental validation for miRNA targets involved in the occurrence and development of bone metabolic diseases in the natural product library.”
Professor Baoting ZHANG’s research team has constructed a knowledge-based calculation (machine learning algorithm learning from established virus nucleic acid and the interaction of small molecules) to predict small molecules targets with loop (miRNA and its complementary mRNA) and AGO protein. Moreover, the research team further applied structure-based calculation to predict small molecule prediction results and the corresponding binding sites based on the binding free energy. Using a series validation experiments, the research team obtained two small molecules (OB-4 and OC-3) from the vast of traditional Chinese Medicine and natural products database, which can target miRNA214-ATF4-AGO complex and miRNA214-TRAF3-AGO complex. They further applied the osteoblast targeted delivery system to deliver OB-4 to specifically target osteoblasts to resist the phenotype of reduced osteogenic capacity of the osteoblast-specific miR-214 transgenic mice, and the osteoclast targeted delivery system to deliver OC-3 to reverse the bone phenotype of osteoclast specific miR-214 knock in mice with enhanced bone absorption capacity.
Dr. Zhenjian Zhuo, a team member of Professor Baoting ZHANG said: “The interesting part is that a single miRNA (miRNA214) could cause a huge difference in the structure score of virtual screening when is applied to different mRNAs (ATF4 mRNA in osteoblast and TRAF3 mRNA in osteoclast), indicating that the virtual screening selection strategy in the natural products library possess high specificity and high efficient power to boost drug discovery in targeting miRNA.”
This research is of great significance in advancing in miRNA targets from basic research to the targeted drug development in the pathophysiology of orthopedic diseases. The research team is now working on to develop a free access webserver based on the virtual drug screening for miRNA targets to enable other researchers to use. In addition to that, the research team is now advancing the obtained small molecules from basic research to clinical application.
This work has accepted widely concerns and highly appraises by academic circles. Professor Jiang Hualiang, academician of Shanghai Institute of Materia Medica, Chinese Academy of Sciences, comments on the work: The study provides a virtual screening strategy for drug discovery based on miRNA, just as modern Shennong "tasted hundreds of herbs" in the vast natural products of TCM sources. Professor Xu Hongxi, chairman of Chinese Society of Traditional Chinese Medicine Experimental Pharmacology and dean of School of Pharmacy, Shanghai University of Chinese Medicine, comments on this work: This study provides a highly effective predictive tool for elucidating the novel molecular mechanisms that may exist in the treatment of diseases by natural products of traditional Chinese medicine.
Professor Lu Aiping of Institute of Integrated Bioinformedicine and Translational Science, HKBU, and Professor Zhang Ge of Law Sau Fai Institute for Advancing Translational Medicine in Bone and Joint Diseases, HKBU contributed significantly to this work.
Publication:
Zhuo Z, Wan Y, Guan D, Ni S, Wang L, Zhang Z, Liu J, Liang C, Yu Y, Lu A, Zhang G, Zhang BT. A Loop-Based and AGO-Incorporated Virtual Screening Model Targeting AGO-Mediated miRNA-mRNA Interactions for Drug Discovery to Rescue Bone Phenotype in Genetically Modified Mice. Adv Sci (Weinh). 2020;7(13):1903451.